
Just a decade after Crispr was invented, the first drug to make use of the revolutionary gene-editing technology will be with regulators later this year, promising to transform the treatment of genetic diseases.
In 2012, Nobel Prize winners Jennifer Doudna and Emmanuelle Charpentier published a scientific paper showing that a key part of the bacterial immune system could be used to cut DNA: to alter, remove or correct genetic errors.
Their discovery started a race for new companies to create transformative, and possibly curative, treatments that have developed far more quickly than previous advances in biology.
Nobel Prize winner Jennifer Doudna co-published a paper in 2012 showing that a key part of the bacterial immune system could be used to cut DNA © Innovative Genomics Institute, UC Berkeley
The first to come close to success is Crispr Therapeutics, which, in partnership with biotech group Vertex, hopes to submit late-stage trial data to seek approval of its treatment for sickle cell anemia and the hereditary blood diseases beta thalassemia in the EU and UK regulators at the end. of 2022.
Their latest data show that all sickle cell patients were symptom free and that the vast majority of beta thalassemia patients were no longer dependent on transfusions.
Doudna said it was “extraordinary” that sickle cell patients like Victoria Gray, who was treated in the first trial three years ago, had already been “effectively cured of the genetic disease”.
“One of the most surprising and exciting things for me is how quickly it has been possible to apply it to the clinic,” he told the Financial Times. “It has been the foundation of several companies, created tens of billions of dollars in value and many, many thousands of jobs.”
Consequently, large pharmaceutical groups such as Pfizer, Bayer and Regeneron have taken note, forming partnerships with the pioneers. Analysts predict that drugmakers will soon be in the market to buy Crispr companies, in part in hopes of acquiring the tool to improve breakthrough cancer treatments.
But the development of Crispr also raised important questions about the ethics of gene editing and its long-term effects.
Many scientists condemned a Chinese researcher who revealed in 2018 that he had edited the genetic code of newborn twins. There have also been intellectual property lawsuits, debating the relative contributions of Doudna and Charpentier, and Feng Zhang of the Broad Institute, who wrote a paper showing how Crispr works in humans.
For startups working on Crispr drugs, the general slump in biotech stocks, some failed trials and safety concerns in the adjacent field of gene therapy have made life more difficult. It has become more difficult to convince investors that this revolution has staying power.
And when the first Crispr drugs are approved, the question of persuading insurers and governments to pay the high prices comes up, just as drug price reform is back on the agenda in the US, the pharmaceutical market most great in the world
 The technology is being used to treat inherited blood diseases such as sickle cell anemia © BSIP SA/Alamy
The technology is being used to treat inherited blood diseases such as sickle cell anemia © BSIP SA/Alamy
Biotech “transistor” moment
Vijay Pande, general partner at venture capital firm Andreessen Horowitz, compares Crispr to the invention of the transistor, which revolutionized computer design.
“We’re in the early stages. I think where we are now is maybe similar to the 1960s in computers,” he said.
The biggest remaining scientific challenge is to deliver Crispr to other organs, beyond the relatively easier areas of the blood, liver and eyes. Kiran Musunuru, a gene editing specialist at the University of Pennsylvania, said researchers are already starting to make progress.
“It’s going to explode in the next 10 years,” he said. “This will be transformative because it will greatly expand the number of diseases we can treat patients with Crispr.”
Crispr’s scientific success is in part because it can use or adapt technology developed and applied in gene therapy, a method of delivering missing genes to patients that has been used to treat life-threatening conditions such as atrophy spinal muscular
It has also benefited from the explosion of data on the human genome, which helps scientists understand which genes to target.
“What’s so remarkable about Crispr is that the timing was right,” Doudna said.
Scientists quickly built on the original discovery. About five years ago, they created the base edition, which can make more precise changes. Pfizer has partnered with base-editing company Beam Therapeutics in a deal worth up to $1.35 billion, hoping to deploy its expertise in lipid nanoparticles, developed from its work on the Covid-19 vaccine, as a way of offering Crispr treatments.
While most trials are still aimed at rare diseases, the base-editing company Verve recently treated its first patient in a study targeting a genetic disease that causes high cholesterol, which could one day be used in a market of millions of patients.
Crispr is also being used to improve a type of personalized cancer treatment called CAR-T therapy, which could be what tempts drug companies to acquire smaller specialty biotechs, according to Jefferies analyst Maury Raycroft.
Big pharma is sitting on huge piles of cash, and some of them need to build their drug pipelines before major patents expire at the end of the decade.
John Leonard, chief executive of Intellia, which first delivered Crispr into the body (rather than removing cells, modifying them and returning them) as part of its trial to treat liver disease , said it’s “essentially a foregone conclusion” that every big pharma company will need to have some Crispr capability.
Healthy skepticism
Not everyone is convinced. After two buoyant years during the pandemic, the entire biotech sector is suffering. Generalist investors are less focused on healthcare, and specialists are concerned that too many companies are going public too soon, often without clinical data.
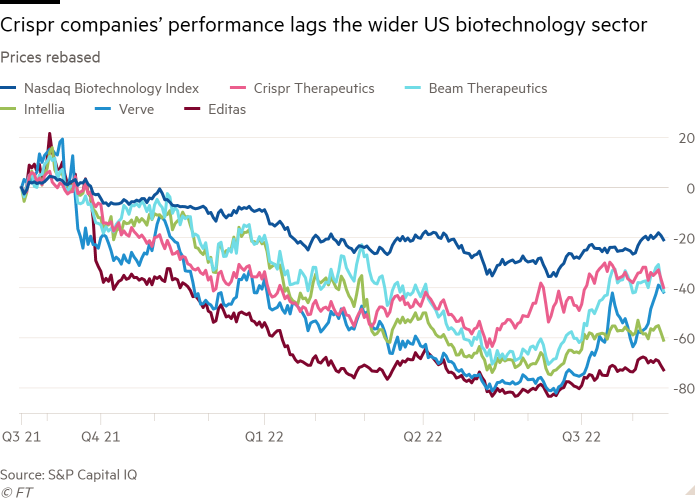
The Nasdaq biotech index fell 21 percent last year, but Crispr companies are down even more. Intellia shares are down 61 percent in the past year, Verve is down 42 percent, Editas shares are down 73 percent, and Beam Therapeutics and Crispr Therapeutics have lost about 40 percent of their market capitalization each.
 A researcher observes a Crispr process. Recent setbacks for gene therapies, though not Crispr specifically, have shaken the market © dpa picture alliance/Alamy
A researcher observes a Crispr process. Recent setbacks for gene therapies, though not Crispr specifically, have shaken the market © dpa picture alliance/Alamy
Nooman Haque, managing director of Emea life sciences and healthcare at specialist lender Silicon Valley Bank, said 2019 and 2020 were record years for the sector, and in particular for gene therapies, but now investors realize there are still challenges ahead.
“There’s a realism that while the technology may be wonderful and amazing, the hard reality of the business is what am I going to get paid for it? Am I going to be substantially better than the competition and in what particular patient group?” he said
The market has also been rocked by several setbacks in gene therapy clinical trials, though not specifically with Crispr.
In 2020, the US Food and Drug Administration rejected a hemophilia therapy from BioMarin, asking for more data, although it has since received conditional approval in the EU. More recently, VBL’s gene therapy for ovarian cancer did no better than existing standard care, while Biogen scored more than $500 million after two gene therapy trials failed.
Even more worrying for some investors were tests halted due to security concerns. Last year, the FDA halted several trials over fears that the therapies could increase the risk of cancer. The highlights were two from BlueBird Bio. A trial was restarted after one participant’s case of leukemia was found to be unrelated to the drug. But in another, a link was established, even though the regulator’s advisers have concluded that the drug’s benefits still outweigh the risks, and it is awaiting approval to restart.
The Crispr companies stress that their technology is significantly different from conventional gene therapies, which involve delivering, rather than editing, a gene. John Evans, chief executive of Beam Therapeutics, said that in some types of gene therapy you can’t always control where the gene is added, but with Crispr you can, and base editing, Beam’s specialty, is even more accurate.
Still, some scientists would like to wait for more data on how the Crispr edits affect patients in the long term.
Luigi Naldini, a gene therapy pioneer at Vita-Salute San Raffaele University in Milan, said overall the outlook for Crispr is “positive” but believes companies should scrutinize the data for a longer period long
“I don’t want to be a Cassandra, but in gene therapy, we have previous experience, follow-up data for a long time. Genome-wide adverse events don’t appear instantly,” he said.
There was good news for gene therapies this week when Bluebird Bio’s treatment for beta-thalassemia received FDA approval. It has a US list price of $2.8 million, making it the most expensive single-dose drug ever.
Evans notes that investors appear to be differentiating between the two areas: Shares of companies using gene therapies that rely on a common delivery method with lentiviral vectors have fallen much more than Crispr companies.
“It’s been kind of a story of two sectors within medicine, between gene therapy, which so far is still kind of reduced to dealing with technical issues, safety issues. and then gene editing, which I think is still it’s bullish, in fact, despite the volatility,” he said.
Gene therapies may have provided key building blocks for Crispr, but they did not solve the problem of how to pay for potentially unique curative drugs. Drugmakers argue that the high prices – Novartis’ unique gene therapy Zolgensma to treat spinal muscular atrophy has a US list price of $2.1 billion – are good value because they save years of healthcare costs for to seriously ill patients.
Steve Seedhouse, an analyst at Raymond James, said the industry is discussing how to make high prices more acceptable, including spreading payments over five years or charging only if a treatment works. “It’s not entirely clear in the long run whether the high price itself will be palatable,” he said.
If its sickle cell treatment is approved, likely next year, Crispr Therapeutics will be the first to test the market for a Crispr drug. Samath Kulkarni, chief executive of Crispr Therapeutics, said that this and Vertex will “set the foundation for a whole new class of drugs.”
“Ultimately, it will change the face of the industry,” he said.
[ad_2]
Source link


